طرز تهیه ی نایلون 6و6
This page is all about how to make nylon. We're telling you this as if you can't read the title for yourself. To start off, nylon is made by a reaction which is astep-growth polymerization, and a condensation polymerization. Nylons are made from diacids and diamines. If you want to see what adipic acid and hexamethylene diamine look like in 3-D, click here.

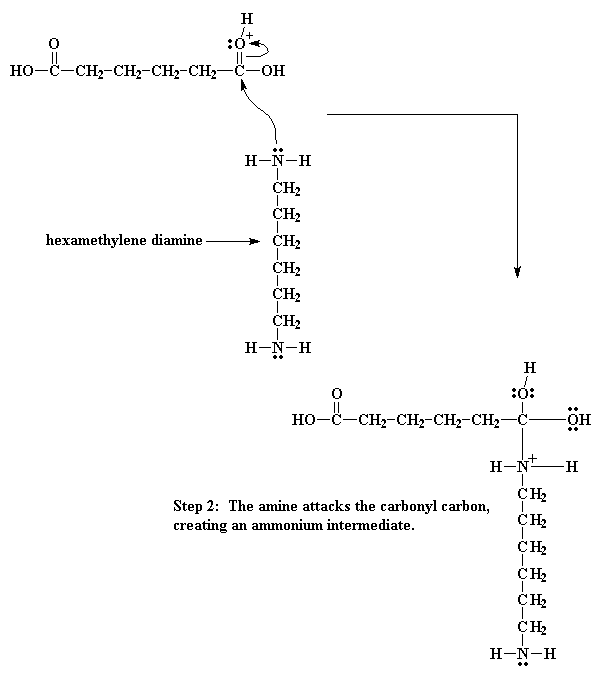
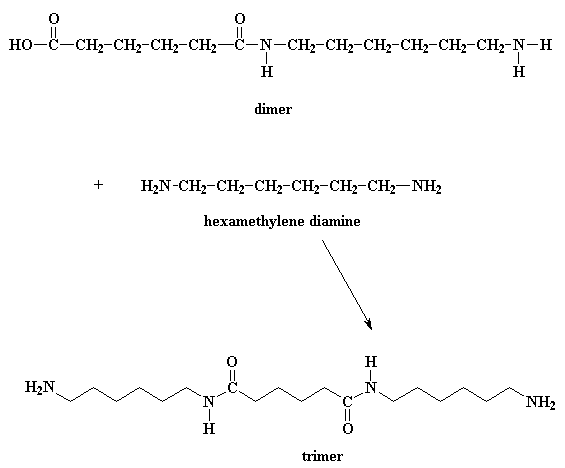
To make nylon 6,6 on doesn't need a catalyst, but acids do catalyze the reaction, and wouldn't you know it, one of the monomers is itself an acid. A little reaction happens between two adipic acid molecules. One will donate a proton to a the carbonyl oxygen of another When this oxygen is protonated the carbonyl oxygen becomes much more vulnerable to attack by the nitrogen of our diamine. This happens because our protonated oxygen bears a positive charge. Oxygen does not like to have a positive charge. So it pulls the electrons it shares with the carbonyl toward itself. This leaves the carbonyl carbon lacking electrons, and ready for the amine nitrogen to donate a pair to it Then the electrons play musical chairs. One of the electron pairs form the carbonyl double bond shifts entirely to the oxygen, taking care of the problem of the positive charge at that atom, but now our nitrogen has a positive charge So then we get an even more elaborate game of electron musical chairs. The electrons from the hydrogen oxygen bond go back to the oxygen, freeing the proton, regenerating the acid catalyst. Then the carbonyl oxygen shares its newly regained electrons with the carbon atoms, regenerating the carbonyl double bond. Of course, this isn't enough. The oxygen of the hydroxyl group decides to do a little bit of electron shuffling of its own. It takes the pair it shares with the carbon and hogs them to itself, severing the bond between it and the carbon. It then donates a pair of electrons to a hydrogen connected to the nitrogen. That gets this hydrogen thinking. As it now shares a pair of electrons with the oxygen, it sees no need to keep the pair it shares with the nitrogen, so it lets go of that pair, giving it over to the nitrogen. This shift of electrons breaks the bond between the hydrogen and the nitrogen, and gets rid of the positive charge on that nitrogen. It splits off H2O, and generates the amide-containing dimer. So what does this dimer do? Look close, and you'll see that it has an acid group at one end, and an amine group at the other. This means that it can react with a molecule of the diacid, or a molecule of the diamine. Either way, you get a trimer Wanna know a little secret? Our dimer can also react with other dimers, to make a tetramer if it wants to. Or it can react with a trimer to form a pentamer, and it can also react with bigger oligomers. Eventually, when this happens, dimers will grow into trimers, tetramers, and bigger oligomers, and these big oligomers will react with each other, to form even bigger oligomers. This keeps happening until they become big enough to be called polymers



 نوشین خلقتی هستم .
نوشین خلقتی هستم .